Biocon Gives Clarification on Clinical Study of Itolizumab for COVID-19 after Backlash - IndiaMedToday
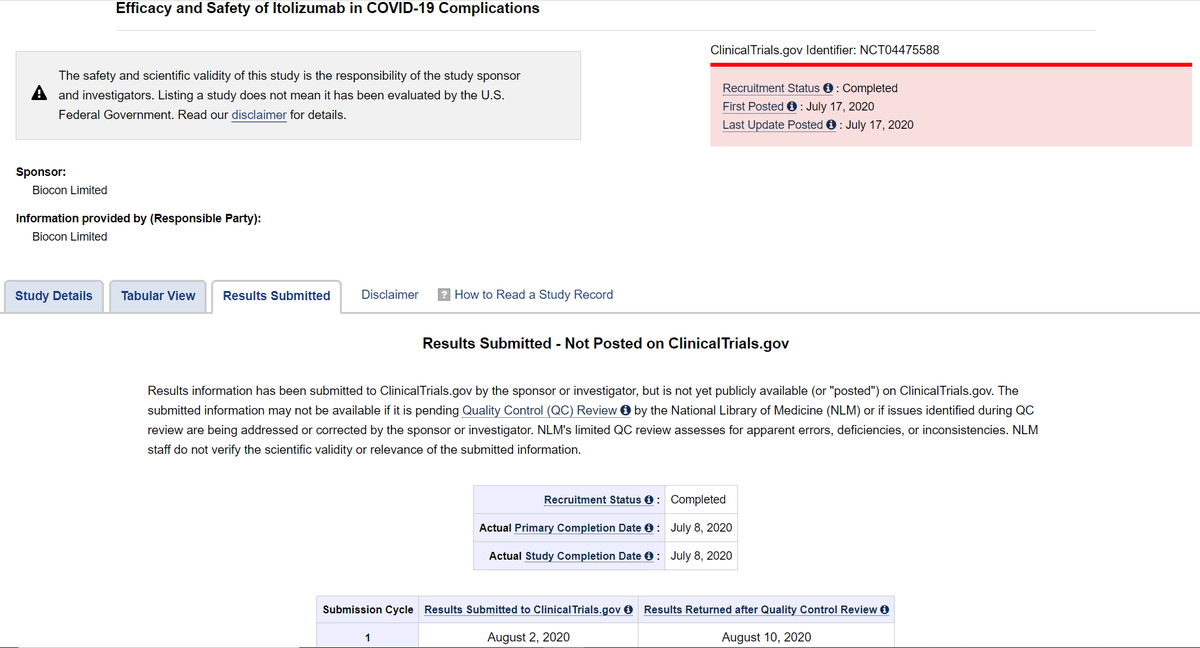
सिद्धार्थ on Twitter: "Brief summary here: 👉 Biocon Biologics had submitted result to https://t.co/pXvPY9Oaef @NIH on Aug 02, 2020 BUT results were returned after quality control review on Aug 10, 2020. On

Infographic: Biocon opens this year with a stellar performance reporting 71% growth in profits | AlphaStreet

Strategic Management Analysis of Biocon India Group Limited - CIRCLE OF BUSINESS Strategic Management Analysis of Biocon India Group Limited

CBI arrests Joint drug controller for allegedly taking Rs. 4 lakh bribe to clear Biocon Biologics's diabetes injection; company denies | The Financial Express

Biocon's coronavirus drug for emergency use given approval after trial on only 30 patients - India News

Biocon Biologics enters strategic collaboration with Serum Institute unit - Pharmaceutical Technology

Biocon's Itolizumab Drug For Emergency Use on COVID-19 Patients Gets Approval After Clinical Trial on 30 Patients Across Only 4 Hospitals | 📰 LatestLY