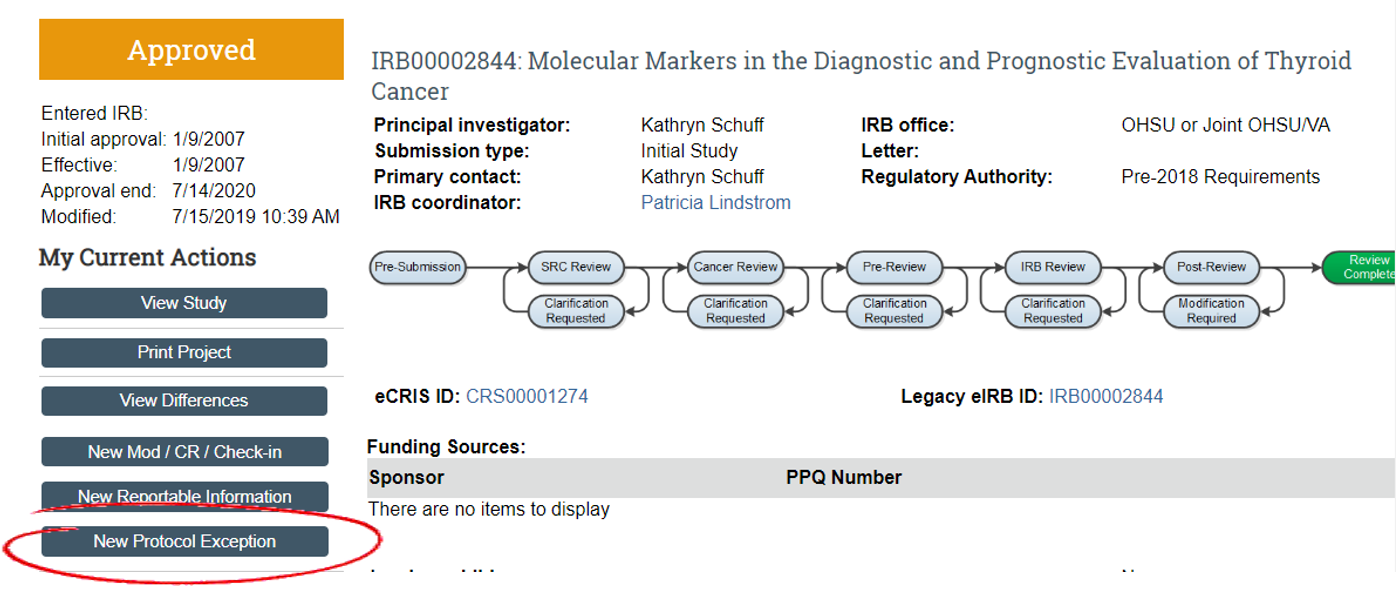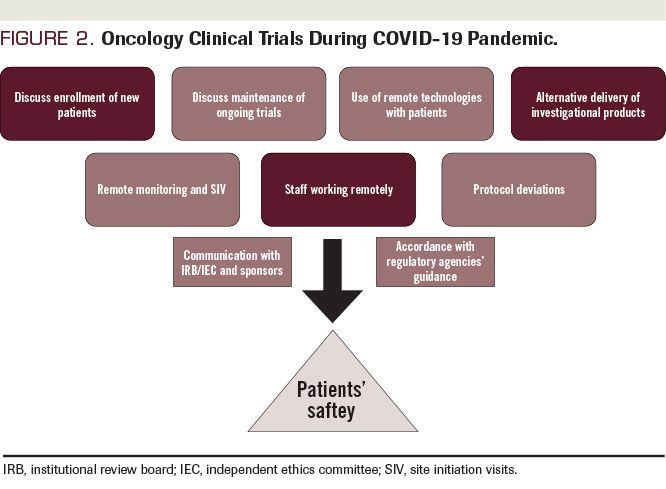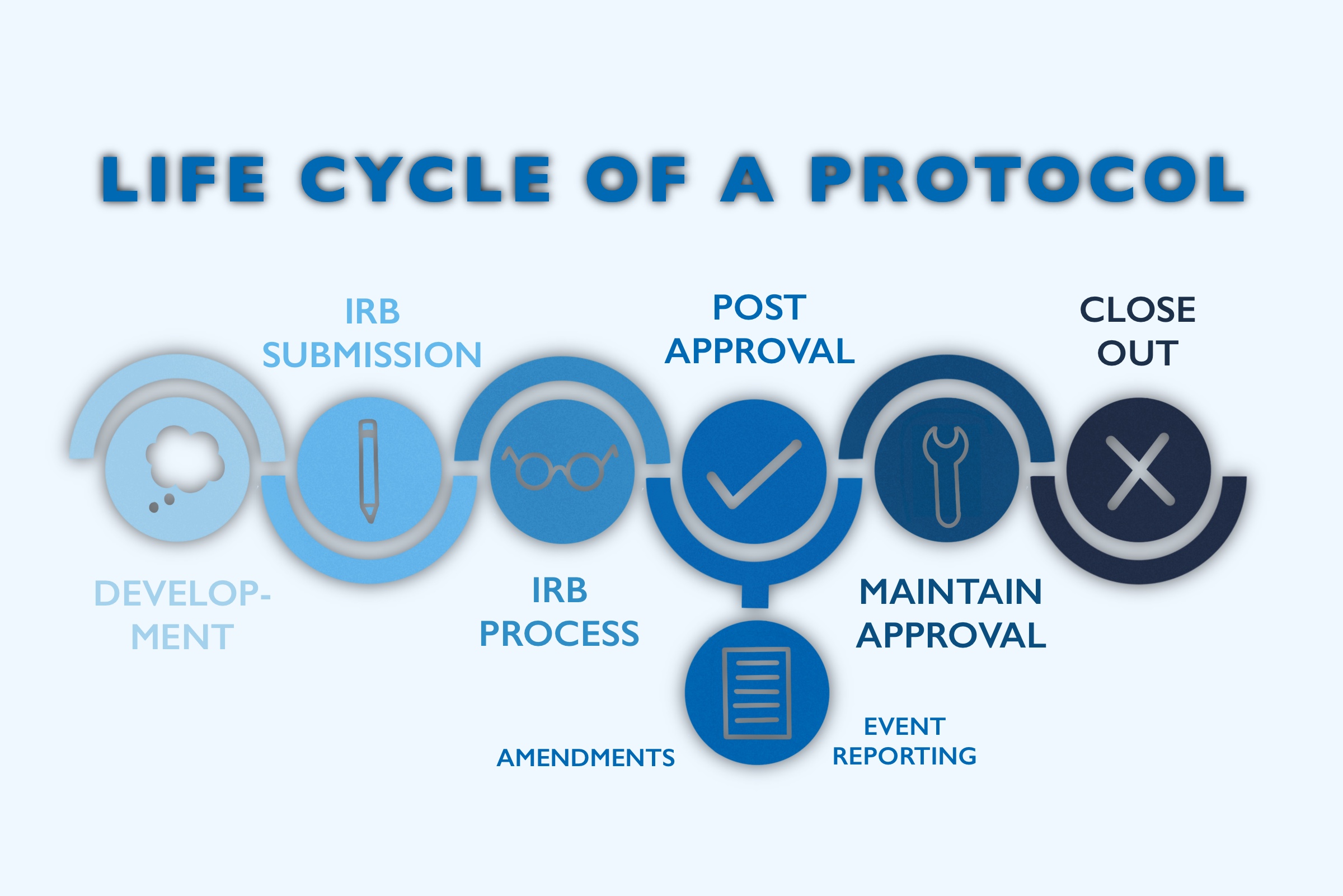
How do Institutional Review Boards (IRB) and Ethics Committees (EC) impact clinical trials? - Clincierge

INSTITUTIONAL REVIEW BOARD : A REFERENCE GUIDE: 9781686777486: Medicine & Health Science Books @ Amazon.com

Main Line Health - Every clinical trial conducted in the US is approved and monitored by an independent Institutional Review Board (IRB) to make sure the risks are as low as possible.
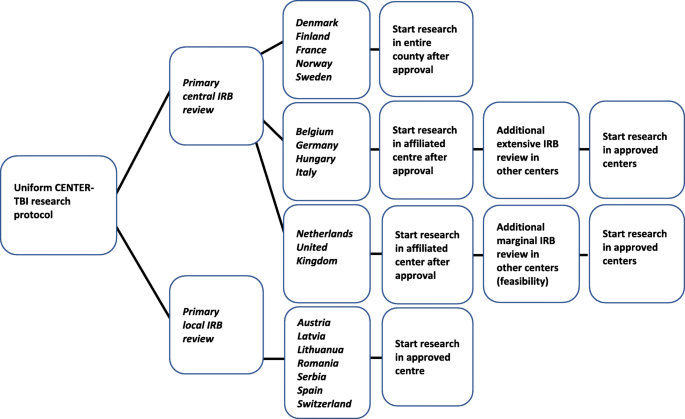
How do 66 European institutional review boards approve one protocol for an international prospective observational study on traumatic brain injury? Experiences from the CENTER-TBI study | BMC Medical Ethics | Full Text