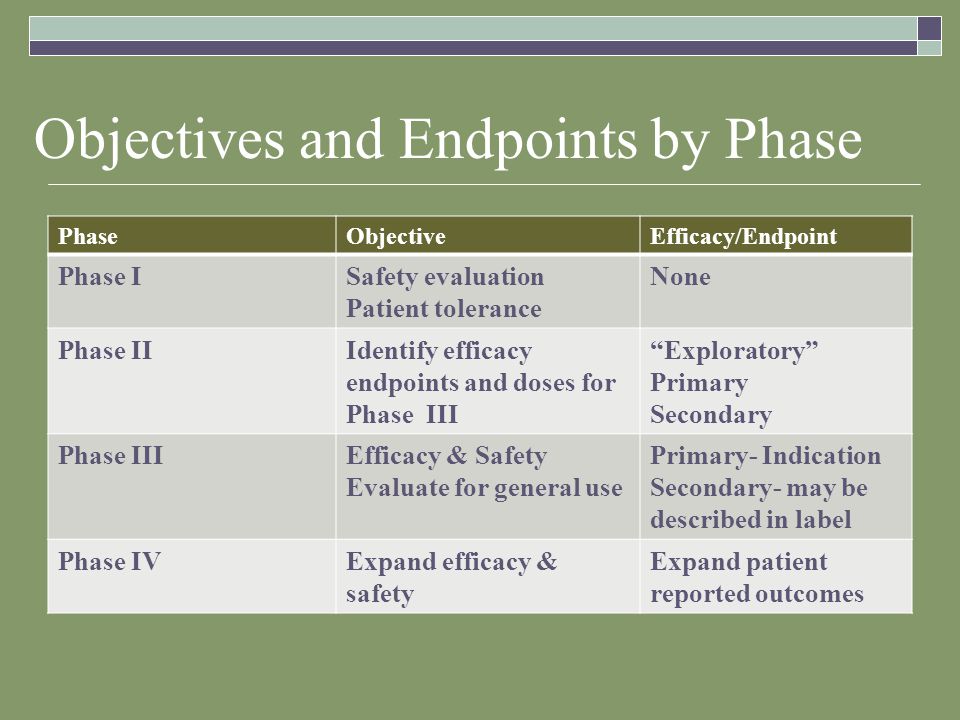
Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews
![PDF]❤DOWNLOAD⚡ Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines - Flip eBook Pages 1-1 | AnyFlip PDF]❤DOWNLOAD⚡ Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines - Flip eBook Pages 1-1 | AnyFlip](https://online.anyflip.com/hxgrd/dcmn/files/mobile/1.jpg?1624012975)
PDF]❤DOWNLOAD⚡ Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines - Flip eBook Pages 1-1 | AnyFlip
On Biostatistics and Clinical Trials: Control for Type I Error (or Adjustment for Multiplicity) for Secondary Endpoints

Challenging Issues in Clinical Trial Design: Part 4 of a 4-Part Series on Statistics for Clinical Trials - ScienceDirect

Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines by Tom Brody PhD | NOOK Book (eBook) | Barnes & Noble®
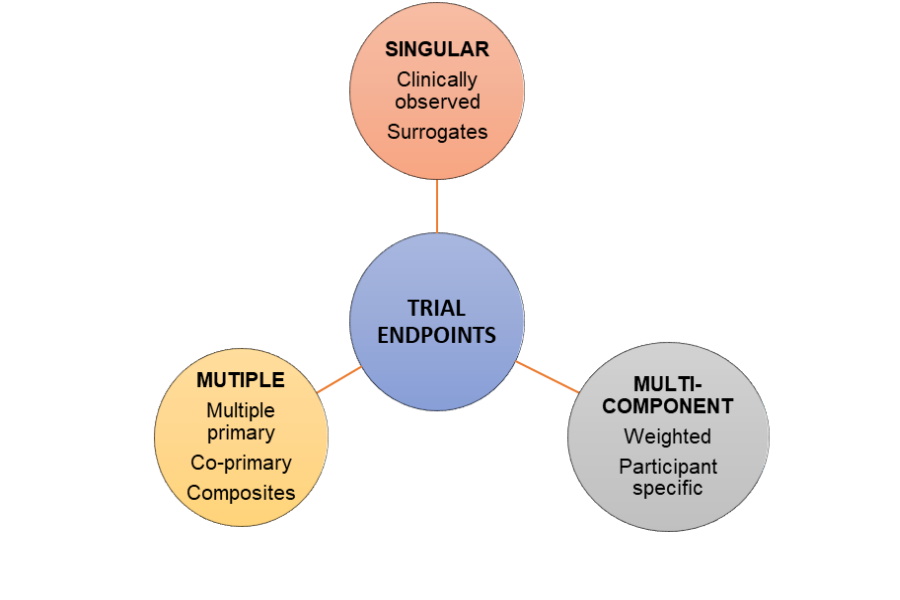
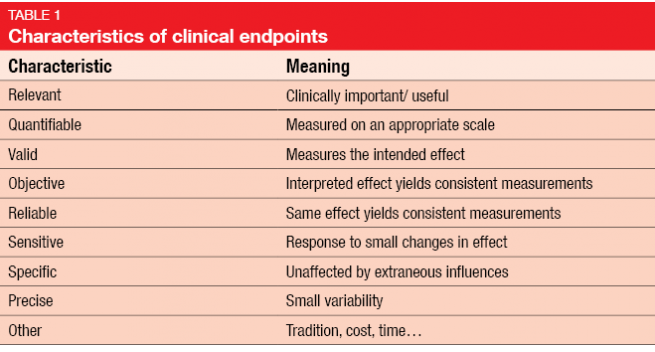

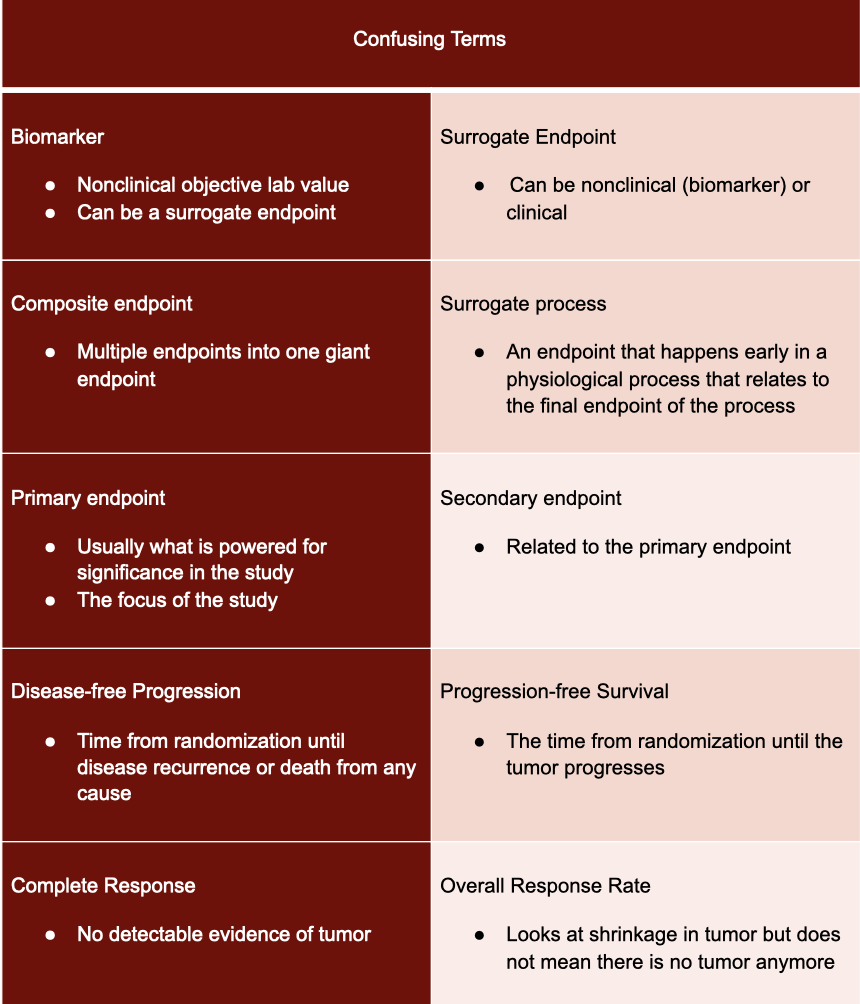








![Stages of PWS Drug Development Overview [VIDEO] Stages of PWS Drug Development Overview [VIDEO]](https://www.fpwr.org/hubfs/Clinical%20Trials/Drug%20Development%20Process/Slide5.jpeg)